How to find and take part in clinical trials?
You might question, why should I try something that researchers are unsure will work or why should I participate in clinical trials near me. That is a valid question. Clinical Trial participation may have risks, but it also has its advantages. Many people are hesitant to participate in research because of past Clinical Trial experiences. Today, though, there are strict laws in place to protect your privacy and physical well-being.
The results of Clinical Trials can advance the knowledge of diseases and help in detection, diagnosis, and care. Revival Research Institute is one of those platforms that provide Clinical Trials in Michigan. We strive to provide the highest caliber of care to our patients, driven by the excellence of our experts.
Our skilled medical staff and professionals are dedicated to offering the best patient care while upholding the objectives of each study. To bring innovative and game-changing treatments from the lab to patients, Revival Research Institute works with brilliant minds from a variety of therapeutic fields.
What are clinical trials?
Clinical Trials occur when a group of patients is subjected to a new medical procedure, drug, device, or method to see how well it works. Finding a novel and improved method to treat, prevent, or diagnose various illnesses is the goal of a Clinical Trial.
These novel treatments are initially tested in laboratories by scientists. Then they test them on lab animals. A Clinical Trial on humans won’t begin until they decide the potential therapeutic approach is safe and effective enough in these early stages. Small groups will be tested first, followed by larger ones in phases.
If you have a severe illness and have run out of effective standard treatment options, it could be especially helpful to participate in clinical trials near you in Michigan.
What Are the various phases of clinical Trials?
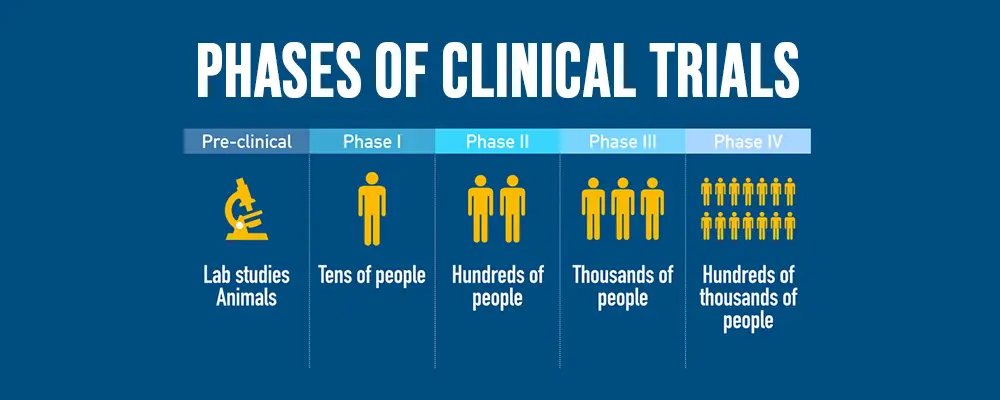
Clinical trials have 4 phases which are as follows:
- Phase I: To test for safety, doctors administer a novel treatment to a small sample of patients. The best way to administer the novel therapy, monitor any potential side effects, and a secure dosage are all determined by the researchers.
- Phase II: The research team looks at how well the medication treats a specific ailment.
- Phase III: The team compares the new treatment to the current standard of care and works to determine how various dosages and treatment combinations affect various populations (e.g., men, women, young, old, and various ethnic groups).
- Phase IV: Here, the treatment is generally only tried on willing patients. The objective is to look for side effects not seen in earlier stages and determine how well the course of treatment performs in the long run. Drug companies are permitted by the FDA to market the therapy at this point.
Clinical Trials near you in Michigan:
Revival Research Institute conducts clinical research trials for a variety of diseases and conditions, such as Preventive treatment for Alzheimer’s disease, skin allergies, Asthma, other infectious diseases, as well as cardiovascular disorders. As a top Clinical Research Organization, our staff members combine compassion, science, and ingenuity to run effective Clinical Trials, boost patient recruitment, and keep volunteers on board.
The Principal Investigator is the Physician in charge of each Clinical Trial. They are responsible for overseeing the care of the patients and the overall research study. A protocol, or plan for the Clinical Trial, is created by the Principal Investigator. The research study’s protocol outlines the procedures that will be followed. Additionally, it has details that aid the Physician in determining whether this course of treatment is appropriate for you or not.
The Principal Investigator is the Physician in charge of each Clinical Trial, and they are responsible for overseeing the care of the patients and the overall research study. A protocol, or plan for the Clinical Trial, is created by the Principal Investigator. The research study’s protocol outlines the procedures that will be followed. Additionally, it has details that aid the Physician in determining whether this course of treatment is appropriate for you or not.
The protocol provides details about:
- The rationale behind the Clinical Trial’s eligibility requirements for participants.
- How many individuals are required for the Clinical Trial?
- What medications or treatments may be administered?
- How the dosage will be administered?
- What medical tests will be performed?
- How frequently data will be gathered from the participants?
You can find out about our trial’s goals, eligibility for participation, locations, and contact information at Clinical research studies.
Impact of clinical trials near you in Michigan at Revival Research Institute:
Revival Research Institute has one mission and that is to improve health & quality of life and lessen illness and disability. Numerous studies on specific diseases as well as more comprehensive analyses as a whole provide evidence of the diverse, long-term effects of the investigational product on doctors and researchers.
In carrying out this mission, we aim to advance health by fostering prevention and treatment and benefit society by fostering improvement in healthcare. And increase knowledge in the biomedical sciences by supporting cutting-edge research and training the biomedical workforce.
Why should I participate in clinical trials?
Participating in Clinical Trials is the foundation of all medical advancements in clinical research. They help us examine novel approaches to avert, diagnose, or treat disease.
New drugs or drug combinations, new surgical techniques or medical equipment, or novel applications of current treatments can all be used as treatments.
Clinical Trials seek to ascertain the efficacy and safety of novel diagnostic or therapeutic approaches. Clinical Trials may also examine different facets of treatment, like enhancing the quality of life for those with long-term conditions.
People who participate in Clinical Trials do so for a variety of reasons. Healthy volunteers claim that they take part to aid others and advance science. In addition to helping others, participants in Clinical Trials with illnesses or diseases do so to possibly receive the most recent treatments as well as extra care and attention from the research staff.
Clinical Trials give many people hope and a chance to contribute to the future development of more effective treatments for others.
What occurs in clinical trials are as follows:
- The study team thoroughly explains the research study and learns more about you.
- You sign an Informed Consent Form after receiving all of your questions answered and decide that you want to participate.
- To make sure you are eligible for the trial, you are screened.
- You arrange a first appointment (known as the “baseline” visit) after being accepted into the trial. During this visit, the researchers perform physical and/or cognitive tests.
- You are put into a treatment or control group at random.
- You adhere to the research study’s protocols with your family and let researchers know if there are any problems or concerns.
- You are welcome to stop by the research site regularly for new assessments (cognitive, physical, or otherwise) and staff conversations.
- The research team gathers data on the intervention’s effects as well as your safety and well-being during these visits.
- Throughout the study, you keep visiting your regular doctor for routine medical care.
How do we select participants for clinical trials?
Following your consent, the clinical research staff will screen you to determine whether you meet the requirements to participate in the Clinical Trial and whether any factors could disqualify you. Physical and cognitive tests could be used in the screening process.
Age, disease stage, sex, genetic profile, family history, and availability of a study partner who can travel with you to subsequent appointments may all be inclusion criteria for a research study. Specific medical conditions or medications that might interfere with the experimental treatment may be included in the exclusion criteria.

To find enough volunteers for a study, a lot of people are usually screened. In most cases, you can only take part in one study at a time. Being rejected from one Clinical Trial does not guarantee rejection from another because the criteria for each study differs.
“Can I be Compensated for Participating in a Research Study?”
This is the most frequent concern people have when considering taking part in a Clinical Trial.
At Revival Research Institute, we conduct some a variety of Clinical Trials near you, in Michigan. We may provide reimbursement for travel and/or time spent on research-related activities. The amount of reimbursement for a clinical trial is unique and is based on a variety of factors, including but not limited to:
- The required number of in-person meetings.
- The required number of phone calls.
- Requirements for keeping diaries or tracking symptoms.
- Examinations/Screenings.
- The condition is under investigation.
- The burden on patients and caregivers.
- The study’s duration.
- The stage of the research (earlier phase studies typically involve more risks).
The Takeaway:
No matter how simple it may be, taking part in a Clinical Trial can still involve time, commitment, travel, and potential risk. Due to this, Revival Research Institute offers compensation for all of its studies. Numerous visits are frequently necessary for studies, and each visit may include a reimbursement. Our Clinical Trials cover a broad spectrum of therapeutic approaches including investigational vaccines, drugs, medical devices, or tests.
Revival Research Institute is completely open and honest about all the necessary study requirements and potential reimbursement. You can inquire about a stipend during your initial phone call with our medical staff or during your in-person screening. To obtain Informed Consent, compensation and details about the study will also be covered.
Revival Research Institute has different locations where studies are being conducted in a variety of therapeutic fields. Visit our study page or give us a call to speak with our Recruitment Specialist if you’re curious to learn more about joining a Clinical Trial near you in Michigan.




